A foundational shift is under way in the global health space, thanks to continuous disruptions and groundbreaking innovations. Legacy organizations are embracing change, testing novel business models, and creating new opportunities. At the same time, new players from technology, consumer products, services, and other sectors are entering the enormous global health marketplace, unburdened by conventional business models.
In this new world of convergence, health systems in both established and emerging markets are seeking new ways to meet the demands of diverse populations. At the same time, it is becoming increasingly difficult to draw lines separating technology, science, and medicine. Information and technology-enabled consumers are also fueling change, disrupting traditional ties between life-science companies, providers, payers, and patients.
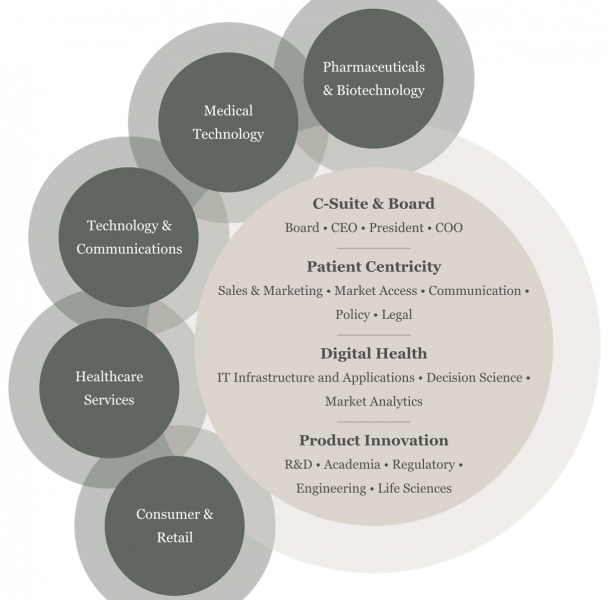
This means that companies need a new kind of leadership. At Egon Zehnder, our Health Practice finds and develops leaders from across industries and geographies with the ability to catalyze change across an entire organization. With our global reach of 63 offices and deep industry experience, we are able to actively advise organizations that are breaking new ground and realigning for this new patient-centric health model.